In the clinic with a young MPN patient
A young adult sits across from Dr. Linda Resar, hands clasped, voice unsteady. The diagnosis is a mouthful — a myeloproliferative neoplasm (MPN) — and feels impossible. As Resar explains, “They go to the internet and they say, ‘Wow, I have cancer! I’m so young! This has to be devastating.’”
She slows the moment down. Her tone shifts. She begins with biology. “I try to educate my patients about their disease and its origin in blood stem cells,” she says. “I discuss the diagnosis in the context of normal blood formation, explaining that all of us acquire mutations, akin to “typos”, in our stem cells as we make blood. In some cases, these mutations land in gene that causes MPN. By taking some of the mystery out of the disease, I hope to calm their fears.”
And when understanding takes root, she sees relief. “I’m thrilled when my patients are doing well. And I also appreciate that they appreciate understanding their disease.”
Her pediatric oncology training shaped this instinct. When working with young people, she thinks in many decades, not years. That mindset guides her MPN care as well. It’s why she tells each newly diagnosed patient, with conviction and hope:
“I want you and your blood stem cells to be healthy for 100 years. That’s my goal.”
That promise is not just poetic; it’s hematopoietic. Every question she asks in the laboratory leads back to the clinic and to the patients seeking answers.
The scientific problem: Why HMGA1 matters in MPNs
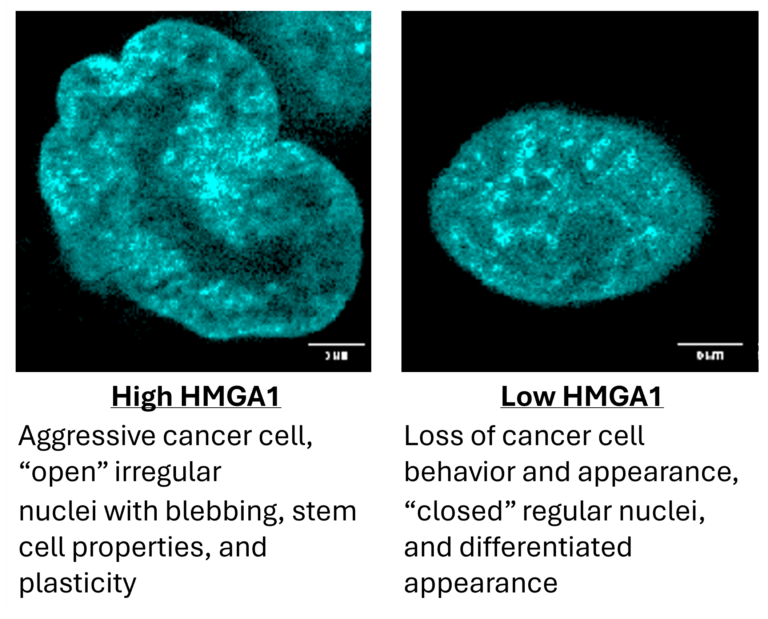
In Resar’s search to understand why some MPNs accelerate while others remain stable for decades, she focused on a family of chromatin‑binding proteins called HMGA. The HMGA family is active in stem cells during early embryo development, when rapid growth is important. When her team began studying this family, “we discovered that they can function as very potent oncogenes, or cancer-promoting genes… Like Goldilocks’ bed, the level of HMGA1 must be perfect – not too little, which would impair blood formation, and not too much, which could cause overproduction of abnormal blood cells.”
The HMGA family of proteins belongs to a group of proteins called transcription factors — molecular regulators that help control which genes are turned on or off. By shaping how genetic information is used, these proteins play a key role in determining how a cell functions and behaves. Unlike traditional transcription factors, HMGA1 binds directly to stretches of the genome rich in the base pairs adenine (A) and thymine (T). HMGA’s are equipped with a special tools, called AT-hooks, that physically bends AT rich DNA regions in unique ways, affecting the 3D structure around particular genes. These bend open and close regions of the genome to “turn on” or “turn off” genes that govern cell growth and behavior.
When a member of this protein family called HMGA1 levels rises, it changes how blood stem cells — hematopoietic stem cells — behave. High HMGA1 levels push hematopoietic stem cells towards embryonic, stem cell-like state, increasing their ability to self-renew in ways that resemble cancer behavior.
HMGA1 opens the genome, activating stem cell programs
As Resar’s group examined HMGA1’s activity, they made a remarkable discovery: when HMGA1 levels rose, stem cell programs switched on, and disease progression followed.
In her words, “We know high levels of HMGA1 occur in mutant stem cells of our patients as they progress. This also occurs in our mouse models as MPN progresses. We also found that if we deplete HMGA1 levels by just 50%, we can prevent progression to myelofibrosis.” Remarkably, mice with 50% HMGA1 are healthy and their lifespan is normal, if not slightly increased.
In MPN models of progression to leukemia, the results were even more striking:
“We discovered that turning down HMGA1 levels prevents the leukemic cells from surviving and expanding in the bone marrow, which is where leukemia cells originate.”
Increased levels of this protein in mutated cells are associated with various forms of cancer. Dr. Resar’s discovery established MPNs on the growing list of cancers HMGA influences.
According to Resar, amplifying HMGA1 “confers a stem-like phenotype causing cells to rapidly divide… and become more malleable or “plastic” which could allow tumor cells to resist therapies.”
HMGA1, then, was directly involved in transforming normal stem cells into aggressive drivers of disease — revealing a molecular lever that could, in principle, be pushed back.
HMGA1 closes the genome, silencing immune activation
The second half of HMGA1’s behavior surprised even Resar’s team. Instead of simply activating genes, HMGA1 could also lock down critical regions of the genome. It was exactly the kind of laboratory moment that makes Resar smile — “ when we get a result that teaches us something completely unexpected.”
Resar explains their intriguing finding: “HMGA is condensing the genome at some of the regions where it binds… it’s closing down regions upstream of genes that make proteins that present foreign invaders or “antigens” to the immune system.”
This discovery pointed toward how MPN cells could perform immune evasion — a subtle molecular tactic that allows mutant cancer cells to hide from surveillance by the immune system.
The implications are profound. If MPN cells suppress their own visibility, therapies that awaken the immune system may need to counter HMGA1’s effects directly. Her laboratory is preparing publications that explore this frontier.
Resar’s vision: Therapeutic possibilities and what comes next
Resar’s research now spans chromatin structure, interferon treatment response, inflammatory signaling, and the search for therapies that nudge malignant stem cells back toward normal behavior.
Her team now uses newer approaches that include single cell transcriptomics and other assays of 3D genome structure at the level of single cells. In Dr. Resar’s words, bulk research methods “average results across all of those cells,” while single cell analyses allow her team to examine what individual cells are doing. This new method reveals what is happening inside individual stem cells “that are going to become megakaryocytes and other progeny that cause problems for our MPN patients.”
By pairing this cell-by-cell view with mouse models that can dial HMGA1 levels up or down, her team can pinpoint exactly which cell types depend on HMGA1 and how this protein reshapes the architecture of the genome.
Their goal: illuminate the physical structure of the genome — the epigenome — of MPN cells and explore the frontier of cancer biology and therapeutics.
Driven to end malignancy
A frequent speaker for MPN patient groups, Resar is quick with a smile and always excited to explain her work. She values curiosity and enjoys the surprises that emerge from the laboratory bench. And she keeps her clinical work front and center, because her research only matters if it improves someone’s future.
Resar also knows the significance of funding from MPN Research Foundation, which supported her early HMGA1 investigations through a 2021 Pilot Progression Grant. That award helped launch new lines of epigenetic inquiry in MPN progression.
Why this work matters now
Every discovery reaches back to the clinic, to the young patients who arrive scared and searching for clarity. Resar meets them where they are, delivers knowledge that restores a sense of control, and returns to the laboratory bench determined to push biology toward a better future.
Her work revealed the HMGA family as master regulators of the MPN genome architecture — proteins that open chromatin to activate stem cell programs and close off DNA access to cripple immune defenses. This dual action reshapes cellular behavior in profound ways. It also reveals molecular keystones where next‑generation therapies could reshape disease architecture.
For Resar, science is never abstract: it’s a direct response to the people she sees in clinic. When she tells a newly diagnosed patient about her centenarian cell health goals, she means it.